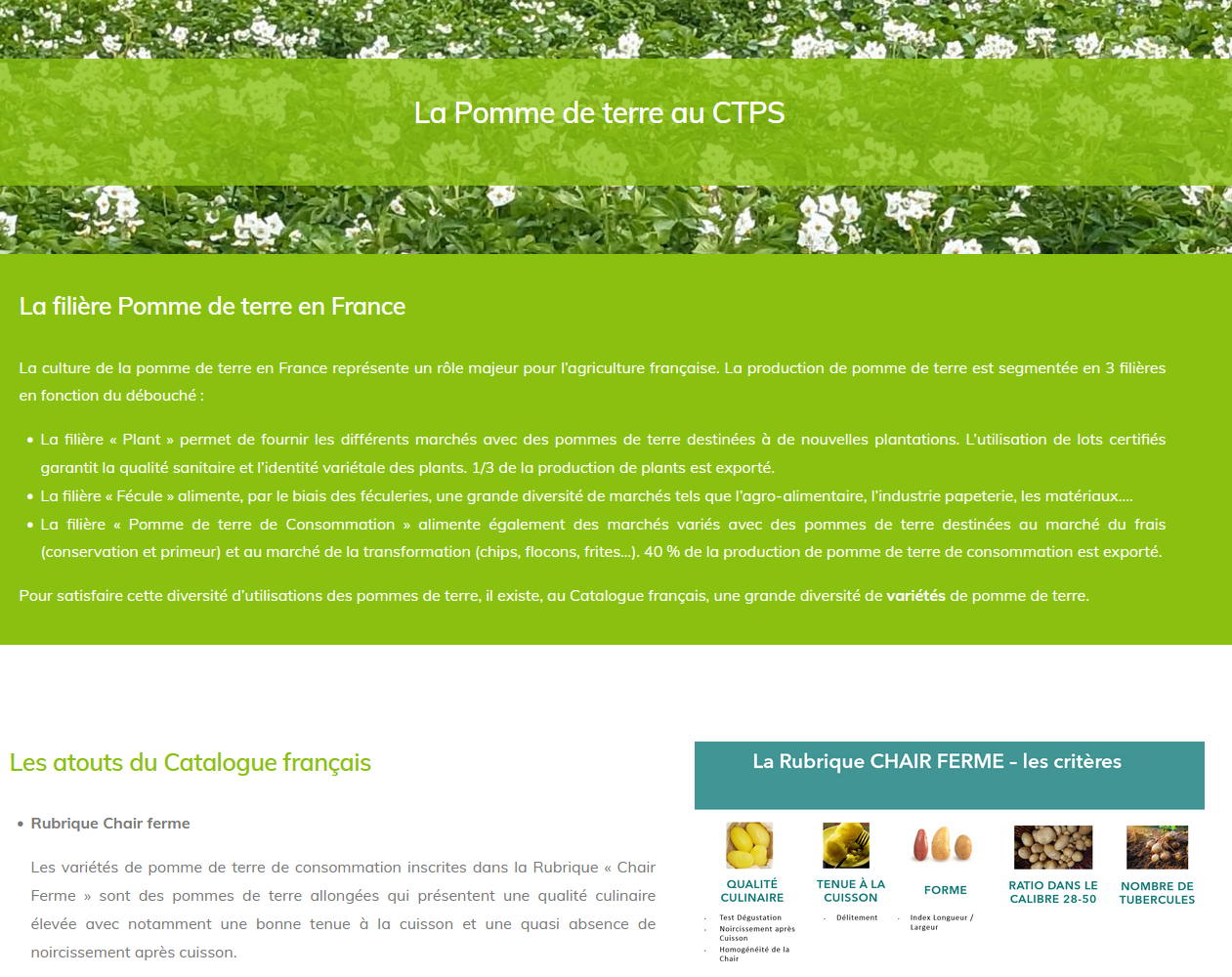
Publication of the CS CTPS referral report on NBT
The European Commission has announced a policy action in 2021 on plants derived from certain new genome editing techniques (NBT). The aim is to adapt authorization and risk assessment procedures as well as traceability and labeling requirements, while maintaining a high level of health and environmental protection.
In this context, in November 2021, the Ministry of Agriculture referred the matter to the Scientific Committee of the CTPS so that it could shed light, on the basis of the scientific and technical literature, on the impact of the evolution of genome editing techniques on the evaluation of varieties and their marketing.
The report in response to this referral was published in November 2022.
Link to the report of the CTPS (French version only)
Link to the summary of the report
It concludes that the use of genome editing techniques does not call into question the major principles of variety evaluation for registration. However, a distinction should be made between edited characteristics that are similar or close to those that can be obtained by conventional breeding, and novel characteristics that are highly modifiable. For the latter, care must be taken to characterize the services that could be provided and the disservices that could appear.
The report emphasizes the difficulty of coexisting varieties derived from NBTs and those not derived from NBTs, due to the limitations of detection. The societal acceptability of these varieties must be taken into account; in particular, it is important that the edited traits benefit the different actors of the sector. The development of the use of NBTs and associated patents reinforces the issues of Intellectual Property related to varieties. The availability of patent information and the use of edited varieties in breeding programs are important issues.